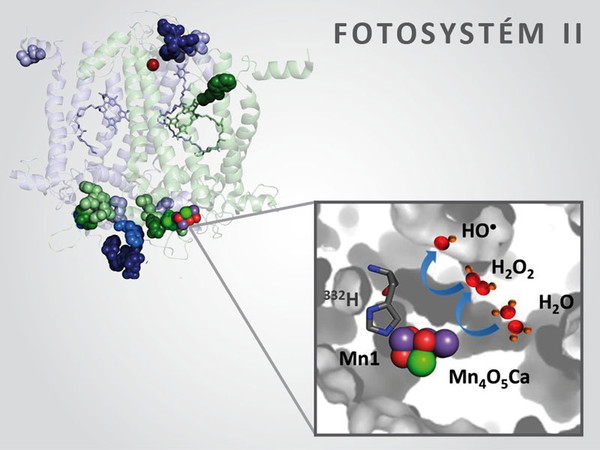
Life on Earth is dependent on molecular oxygen, which is created by the oxidation of water during plant photosynthesis. However, during incomplete oxidation of water, instead of molecular oxygen, hydrogen peroxide is generated, which can create dangerous free radicals. Biophysicists from the Centre of the Region Haná for Biotechnology and Agricultural Research have described this mechanism in co-operation with their American colleagues. The study was published in the prestigious journal PNAS (Proceedings of the National Academy of Sciences of the United States of America).
“The article describes the reaction of the enzyme process created by four atoms of manganese, which is responsible for the oxidation of water and the creation of molecular oxygen during the light reactions of photosynthesis. Water splits into molecular oxygen through four stages of single-electron oxidation, during which free radicals are created. In order to prevent the creation of free radicals, nature has developed an enzyme complex, which gradually accumulates oxidational equivalents for splitting water into four steps and then afterwards water is split at once in four-electron oxidation. The problem takes place if the reaction halts half-way through. Instead of four-electron oxidation, only two-electron oxidation occurs, during which there is the creation of potentially dangerous hydrogen peroxide,” explained the correspondence author, Pavel Pospíšil.
In the presence of transition metals, hydrogen peroxide is reduced into a very dangerous hydroxyl radical, whose existence is counted in nanoseconds. “We are able to capture this radical through the help of electron paramagnetic resonance and study its behaviour in chloroplast membranes,” explained the author of the study, Ravidra Kale.
As a result of the presence of an unpaired electron, a hydroxyl radical has the capability to oxidise all the organic molecules in its surroundings. “We have shown how specifically the hydroxyl radical systematically oxidises the amino acids in two central proteins, D1 and D2, in Photosystem II higher plants,” added Pospíšil.
Olomouc biophysicists have been looking into the problem of free radicals for a long time. The results of the basic research should bear fruit in practise in the future. For example, in ascertaining the higher stability of enzymatic complexes of splitting water for bioreactors, or in artificial photosynthesis, which could introduce a clean energy source for the Earth.